
High-resolution imaging of the change in silicon before and after cycling
The image above shows that the Raman image (color part) of graphite-Si composite electrode is superimposed on the surface profile (gray part) of that. High-resolution component distribution image of nano silicon or ketjen black can be obtained by using RAMANplus with high spatial resolution of 350nm. Here,
(Measurement 1) PVdF is used as binder
(Measurement 2) functional binder PAANa is used
to try to examine the change in cycleability of graphite-Si composite electrode with RAMANplus.
■About the sample used for measurement
Graphite-Si sample:
・Nano silicon powder (Made by Aldrich)
・Natural graphite (grain size 3um)
・Ketjen black
・Binder (PVdF or PAANa)
Electrode composition:
Si : Graphite : KB : binder = 20 : 60 : 10 : 10
Counter electrode, reference electrode: lithium foil
Electrolyte: 1M LiPF6 EC:DMC (1:1)
Cell type: Coin cell (model 2032)
This sample is provided by courtesy of the research group of associate prof. Shinichi Komaba of Tokyo University of Science.
Result 1: isolated inactive nano silicon was observed after volume expansion
The data below is the Raman measured results of graphite-Si composite electrode using PVdF as binder before and after cycling. The high crystallinity nano silicon (red) can be observed on the electrode surface before cycling. But after cycling, apart from the increased alloying part (become amorphous), the conduction to the current collector is prevented by the volume expansion that the nano silicon (bottom left image, inside the red circle) become inactive.
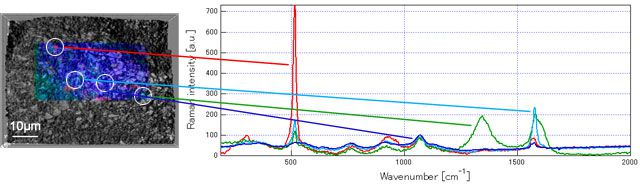
Before cycling: high crystallinity nano silicon dispersed in it
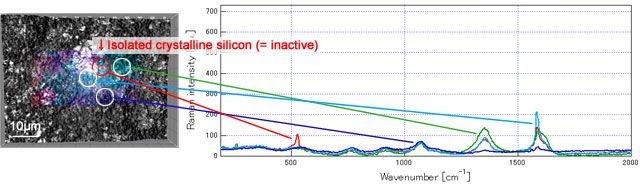
After cycling: nano silicon peels off by the volume expansion and become inactive
Result 2: amorphous silicon alloyed uniformly all over the electrode surface was observed
The data below is the Raman measured results of graphite-Si composite electrode using polyacrylic acid polymer (PAANa) as functional binder before and after cycling. It is reported that the sodium polyacrylate covered the electrode surface can prevent breakdown due to silicon expansion and improve better cycleability. According to the measurements from RAMANplus, amorphous silicon having been alloyed uniformly all over the electrode surface after cycling can be confirmed.
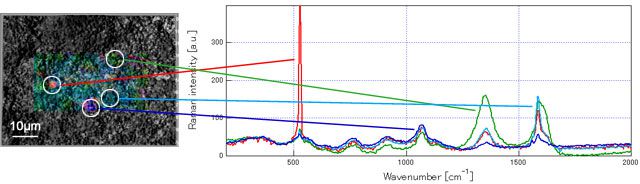
Before cycling: high crystallinity nano silicon dispersed in it
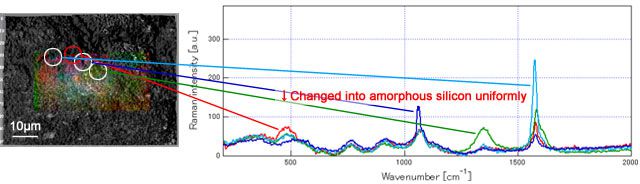
After cycling: amorphous silicon are having been alloyed uniformly all over the electrode surface
(Option) Offer In-situ Raman spectroscopy in inactive gas and electrolyte
Nanophoton can also design a special cell for In-situ Raman spectroscopy to meet every customer’s need. While in the electrolyte, Raman image with higher spatial resolution become available by using immersion objective lens.
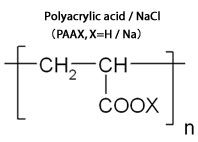
(Reference) About the mechanism of functional binder PAANa
Silicon negative electrode has more than 10 times as theoretical capacity as the conventional electrode and is considered to be the next-generation secondary battery materials. However, in the process of taking in the lithium during charging, the volume expands as much as 4 times that easily result in breakdown. Therefore, cycle life had become short and difficult to put into practical use.
The functional binder sodium polyacrylate (PAANa) uniformly covers the electrode surface and acts as paste that it can prevent the breakdown of silicon particles due to volume expansion. Practical use is expected to be realized because of the performance of silicon negative electrode, the high capacity electrode material, which can be greatly improved by advances in cycleability with PAANa.
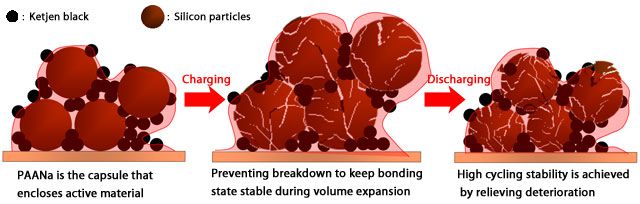
The mechanism of functional binder PAANa
(参考文献)
M. Winter et al., Electrochem. Solid-State Lett.,11, 5 (2008).
S. Komaba et al., Electrochem. Solid-State Lett., 12 107 (2009).
S. Komaba et al., J. Power Sources, 189, 197-203 (2009).
“A powerful new tool that assists in future battery development.”
-Associate Prof. Shinichi Komaba of Tokyo University of Science

“In my opinion, the advantage of Raman microscope developed by Nanophoton is the excellent imaging performance. For instance, what will the change occur in various electrode active material layers painted on a current collector-metallic foil during charge-discharge cycling? How are chemicals distributed? Such information can be obtained by imaging with high-speed and hundreds of nanometer resolution. Excellent method of analyzing crystal structure and surface profile of electrode active material is absolutely essential for realizing high-energy density of li-ion battery. I believe Raman microscope of Nanophoton will become a powerful new tool that assists in future battery development.”