Email Magazine
Free research for adults (8/4/2021)
The days continue to be so hot that I hate to go out. That’s when Mr. Katsuhito Aoki of the Nanophoton Tokyo Showroom told me, “I found a beverage called ice slurry. Ice slurry is a drink made of fine ice grains mixed with liquid, and it effectively cools the body from the inside. Mr. Aoki is my partner in the “free research for adults” project, in which we measure everyday objects with a Raman microscope. How does it look with a Raman microscope that can tell the difference between water and ice? With Mr. Aoki’s help, we immediately tried to measure the difference. (Email Newsletter editor-in-chief / freelance writer Takeshi Nemoto)

According to an article in the Kahoku Shimpo, which also appeared on Yahoo News, ice slurry has long been introduced in professional baseball and other professional sports. When you sweat, your body’s surface temperature drops, but if it is not well regulated, heat will be trapped inside your body, causing your “deep body temperature” to rise. Therefore, to counter the heat, it is important to lower the deep body temperature as well as replenish water and salt. Ice slurry is lower in temperature than liquid and easier to drink, and was proposed overseas around 2010 as an efficient way to cool the body from the core.
I ordered the ice slurry of a famous sports drink as it is commercialized. You freeze the whole plastic pack (100g pack) in the freezer, soften it by hand, and drink it. According to the manufacturer’s website, it was said to be difficult to commercialize, but they have “developed a unique technology to freeze a liquid that can be stored at room temperature and turn it into a slurry. When you drink it at room temperature, it feels like a mixture of melted jelly. I wonder if this is the secret to making ice slurry.
By the way, in order to measure ice slurry, it is necessary to lower the sample to freezer temperature. For this purpose, we borrowed a “Peltier-type cooling and heating stage for microscopes” from Japan High Tech Corporation, which sells specialized equipment. It can be attached to the microscope and lowered to minus 40 degrees Celsius by electronic cooling without using liquid nitrogen. The speed at which the temperature is raised or lowered can be set by you. A person from Japan High Tech visited our Tokyo showroom and set it up on the Laser Raman Microscope RAMANtouch!


Finally, the measurement. First, a spacer was placed between two pieces of cover glass to create a cell to contain the sample. The temperature was changed at a rate of 10°C/minute and kept constant during the measurement. For comparison, we also prepared a conventional sports drink that is not for ice slurry.
First, how does it look when it is frozen? We measured it at minus 20 degrees Celsius.

On the left is a sports drink for ice slurry, and on the right is a conventional sports drink that is not for ice slurry. Both are commercially available. When the Raman spectra of both samples were analyzed, the one for ice slurry did not freeze completely and was divided into two parts: the “ice + sugar” part (light blue) and the “water + ice + sugar” part (red). On the other hand, the conventional sports drink froze almost completely and was divided into an “ice” (light blue) portion and an “ice + sugar” (red) portion. The light blue is the measurement point where the peak characteristic of ice (3140cm-1) appeared, and the red is the measurement point where the peak of sugar (2930cm-1) strongly appeared.
By the way, how can we distinguish between water and ice with Raman microscope? We measured the Raman spectra of water and ice in advance, and the results are shown in the figure below: water at 0°C and 25°C (middle and bottom spectra) have the same peak features, but when frozen at -20°C (top), the peak moves to the left.
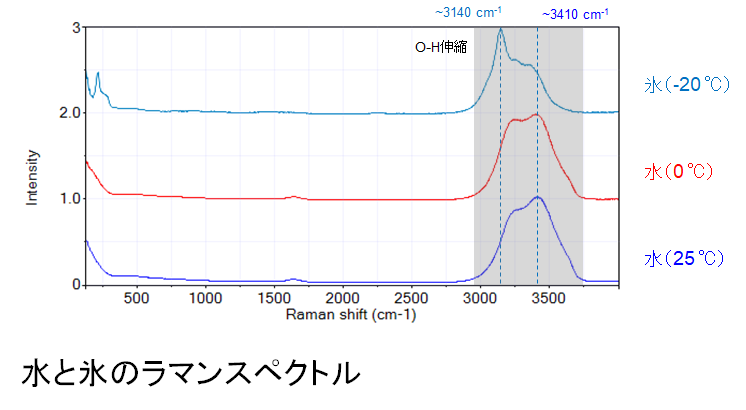
In liquid water, the water molecules move around, while in solid ice, the water molecules are arranged in a regular pattern. This difference affects the vibrations inside the water molecules and appears as a difference in the peaks of the Raman spectra.
Aoki said, “The strong peak appearing around 3000-3700 cm-1 (shaded area) originates from the stretching vibration of OH bonds in water molecules. These OH stretching vibrations are affected by the hydrogen-bonding network, and the spectral shape (vibrational frequency and peak intensity) changes. In liquid water, water molecules are constantly changing their hydrogen bonding partners, while in solid ice, water molecules are regularly arranged and form hydrogen bonds in a stable manner. This situation of water molecules is clearly reflected in the Raman spectrum, so we can distinguish between the different states of matter such as water and ice,” he explains.
Now, back to sports drinks. What does a sports drink look like at minus 5 degrees Celsius?

Sports drinks for ice slurry are found in the form of a slurry of tiny ice grains mixed with liquid. This is probably why it goes down your throat so easily and cools you down from the inside out. Conventional sports drinks, on the other hand, have large, interconnected ice grains.
Let’s take a look at the Raman spectrum obtained by measuring an arbitrary point.

According to the packaging of both sports drinks, the ingredients include sugars such as sugar, dextrose fructose, palatinose, dextrin, and polysaccharides. 2930cm-1 and other peaks other than water and ice are thought to be derived from these sugars.
Mr. Aoki said, “In an ice slurry sports drink, when the ice peak appears, the sugar peak also appears with it, indicating that the ice contains sugar. On the other hand, in conventional sports drinks, no sugar is found in the ice portion. Also, in both sports drinks, when the water peak appears, the sugar peak also appears strongly, indicating that the sugar is dissolved in the water,” he said. Is the secret of ice slurry in the sugars that freeze with the water?
Using a Raman microscope and a cooling/heating stage, we can study various things such as how the size of ice grains changes with changes in composition and concentration. You can also track the freezing and melting of the ice. But this is just a free research project. I’ll leave it at that.
*In the Raman microscopy image shown in the figure, the light blue color is the OH stretching peak of ice around 3140 cm-1, and the red color is the sugar peak around 2930 cm-1.