Email Magazine
The Path to Mastery (10/31/2022)

Do you know what UV curable resin is? My wife purchased a UV-curing resin and a UV light at home, and I was wondering what it was. As the name suggests, it is a gel-like resin that hardens when exposed to ultraviolet light. She uses it to make accessories and for nail art. I looked it up on the Internet and found that it seems to show differences in Raman spectra before and after curing. …… I immediately brought the resin and light to Nanophoton’s Osaka showroom to take measurements. (Takeshi Nemoto, Editor-in-Chief of the Email Magazine / Science Writer)
My wife has made accessories and other items with four-leaf clovers trapped inside. Come to think of it, I had received one before.

When I looked up UV-curing resins, I found an explanation that they contain monomers and oligomers and a photoinitiator. When UV light is applied, the photoinitiator decomposes to generate radicals, which act on the monomers and oligomers, causing a polymerization reaction to proceed. Polymerization proceeds in a few seconds and cures immediately.
The fact that it does not harden unless exposed to UV light, but hardens immediately when exposed to UV light, makes it useful for making accessories and nail polish.
Now, I have several UV curable resins at home, and I borrowed a product that says “Ingredient: acrylic UV curable resin”. The estimated curing time is “UV light 36W, approx. 4 minutes”. The UV light at home was also “36W”.
Since it is acrylic, it seems that radicals generated by UV light attack the double bond (C=C) of acrylate and polymerization begins. if there is a C=C bond, a peak should appear at 1640cm-1 in the Raman spectrum, so we should look at the intensity change.
Now comes the measurement. In fact, the measurement of UV cured resins has already been introduced on the Nanophoton website. The page is titled “Cure degree of Resin and Its Distribution”. Please refer to this page as well.

When I put a drop of UV-curing resin on a glass slide and expose it to UV light, it does indeed harden. This is probably due to the small amount of resin.
I was not sure how I would measure it. In the end, I decided to expose it to UV light for 10 seconds to harden only the surface and measure it from the outside to the inside with a Raman microscope. Hopefully, the outside would be cured and have a smaller C=C-derived peak, while the inside would have a larger peak because polymerization had not progressed.
UV-curing resin was dripped onto white paper and exposed to UV light for 10 seconds. The outside is hardened. When I crushed it with my finger, uncured resin came out from inside.
Finally, it is time for measurement. The measurement mode was set to “XZ Imaging,” which simultaneously measures 400 points in a horizontal row at a time and scans them in the depth direction. The exposure time was 10 seconds, and measurements were taken at intervals of 20 µm from near the top of the sample to 1000 µm down, thus taking less than 9 minutes.
The results were as expected.
The figures below show spectra at depths of 200 μm, 400 μm, and 600 μm from the start of the measurement. Note the third peak from the right.
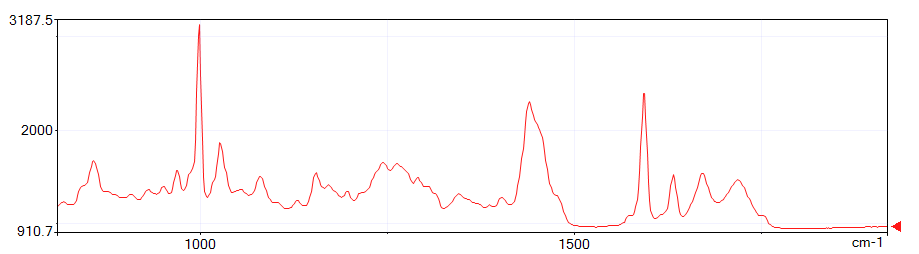
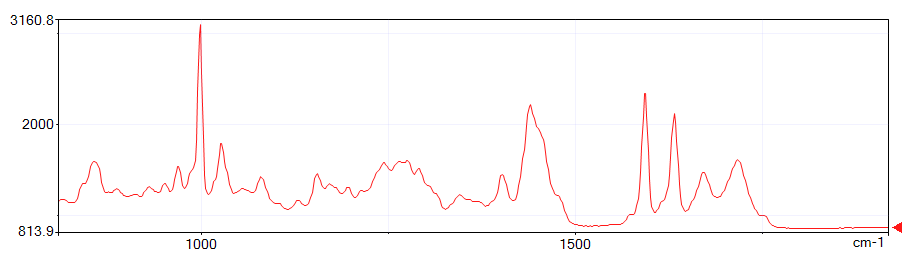
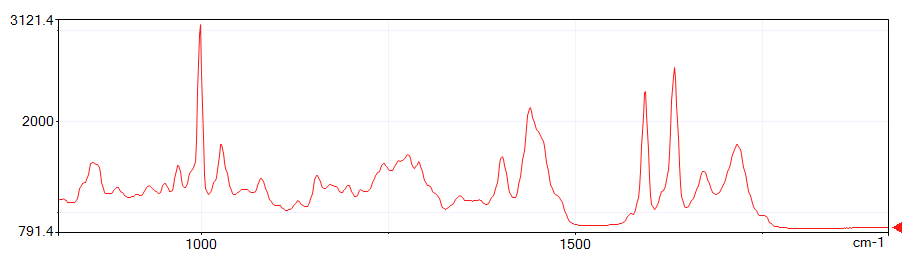
This third peak from the right is the C=C origin.
On the outside of the sample, polymerization has progressed due to exposure to UV light and C=C has decreased. On the inside, polymerization has not progressed, so C=C remains.
I am pleased with myself that this time I got pretty clean data. there are other peaks that are changing besides the C=C derived peak, so I will try to find out what they are derived from.